Isothermal vs. Adiabatic CSTR Esterification: Acetic Acid + Ethanol → Ethyl Acetate + Water Process Simulation in DWSIM.
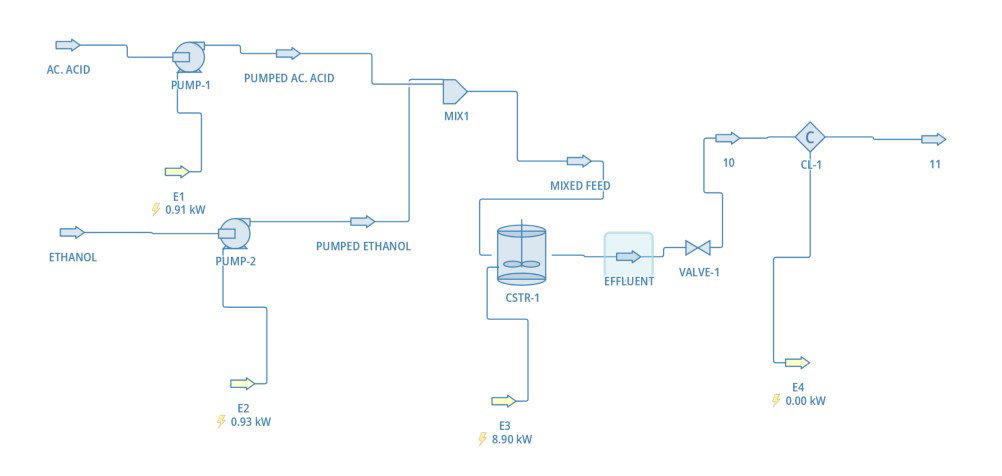
The flowsheet shows our full simulation setup. Two separate feed streams, pure acetic acid and pure ethanol, are each pressurized by their own pump before being combined in a mixer. That mixed feed enters the CSTR where the reaction takes place, then passes through a pressure letdown valve and finally through a product cooler on the way out.
In this simulation, we reacted acetic acid and ethanol in a CSTR to produce ethyl acetate and water, a classic reversible esterification. We ran the reactor under both isothermal and adiabatic conditions to compare how each operating mode affects product yield and whether active temperature control is actually necessary for this reaction system. The key results we captured were effluent composition, reaction rate, reaction heat, and conversion. The main goal was to understand the thermodynamic limits of this reaction and what it would take to push conversion higher at scale. Our feed going into the mixer was a 50/50 equimolar split of pure acetic acid and ethanol, both pressurized by individual pumps before combining. That 1:1 ratio is intentional and important. Any imbalance shifts the equilibrium and cuts into your maximum theoretical yield before the reaction even starts.
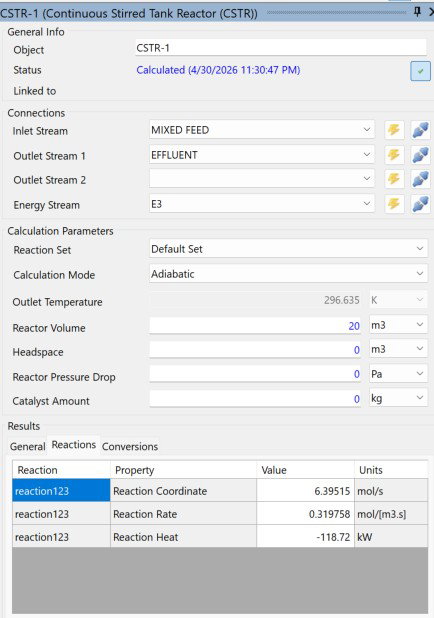
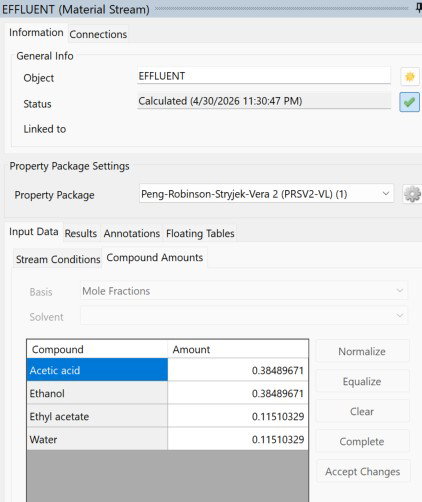
In adiabatic mode, the outlet temperature dropped only about 1.5 K from the inlet, landing at 296.635 K. That tiny temperature swing paired with a reaction heat of 118.72 kW is actually one of the more interesting findings here. It tells us the exotherm is mild enough that the reactor stays thermally stable on its own without any active cooling.
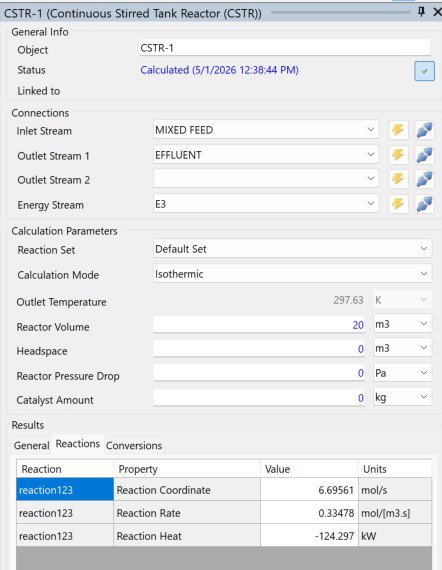
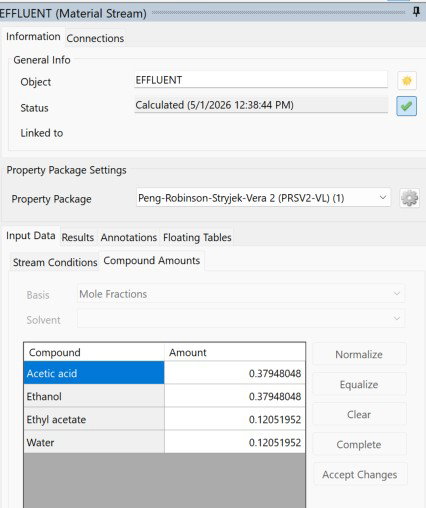
Running isothermal this time at 297.63 K pushed things noticeably further than the previous run. Ethyl acetate climbed from 0.11510 to 0.12052 mol fraction, a small but real gain in product yield. The reaction coordinate increased to 6.696 mol/s and the rate ticked up to 0.3348 mol/(m³s), with the reaction releasing 124.297 kW of heat.
Acetic acid and ethanol are locked at 0.37948 each, and ethyl acetate and water match at 0.12052 each, which confirms the stoichiometry is balanced and the equimolar feed is doing its job.
Overall, the isothermal mode continues to show stable, predictable behavior.
This simulation show that we see a higher yield in the isothermal reaction, but with the tradeoff that more heat is produced which can cause poor reliability of assets.